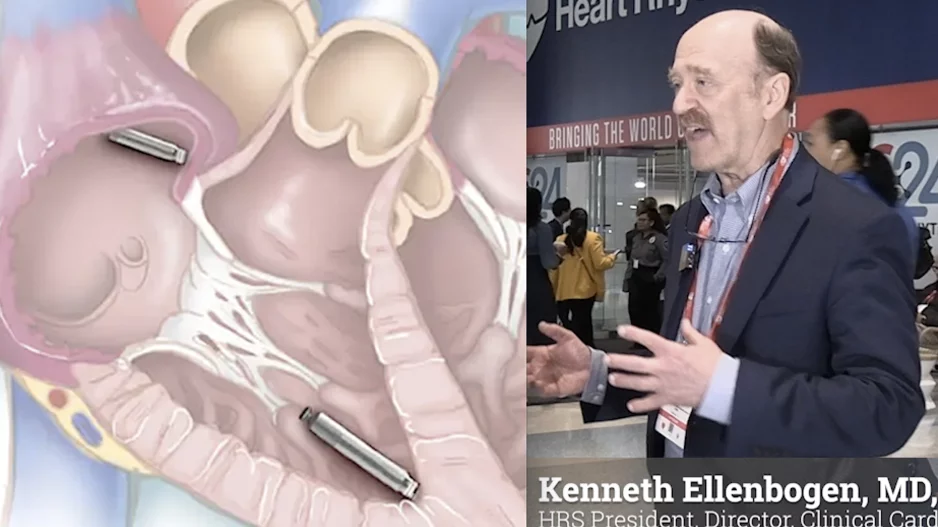
More Articles
Medicare Part B enrollees will pay significantly less for 64 different prescription medications for the next three months, according to a new announcement from the U.S. Department of Health and Human Services (HHS…
Chicago-based Tempus AI has gained clearance from the U.S. Food and Drug Administration (FDA) for a new artificial intelligence (AI)…
Patients with persistent atrial fibrillation (AFib) who undergo transcatheter aortic valve replacement (TAVR) may face a heightened risk of long-term…
Energy drinks may increase the risk of sudden cardiac arrest (SCA) in people with underlying heart disease, according to a new analysis published in Heart Rhythm.[1]
It is too early to know for sure, researchers wrote, but the…
Medicalgorithmics, a Polish healthcare technology company focused on using advanced artificial intelligence (AI) models to diagnose arrhythmias, has received approval…
Elutia, a Maryland-based healthcare technology company, has received clearance from the U.S. Food and Drug Administration (FDA) for EluPro, its new biologic envelope specifically designed for patients with…
Kardium, a Canadian medical device company focused on electrophysiology technologies, has raised $104 million in new financing. Fidelity Management and Research Company led the funding round; T. Rowe Price Associates and new investor Durable…
The U.S. Food and Drug Administration (FDA) is warning Americans not to “eat, sell or serve” mushroom-laced chocolates sold by Diamond Shruumz. Sickness and hospitalizations after eating these products have been…
Stuart Connolly, MD, a veteran cardiologist known for his groundbreaking work in the field of electrophysiology, died June 2 after a long battle with neuroendocrine cancer. He was 75 years old.
Connolly spent 40 years at McMaster…
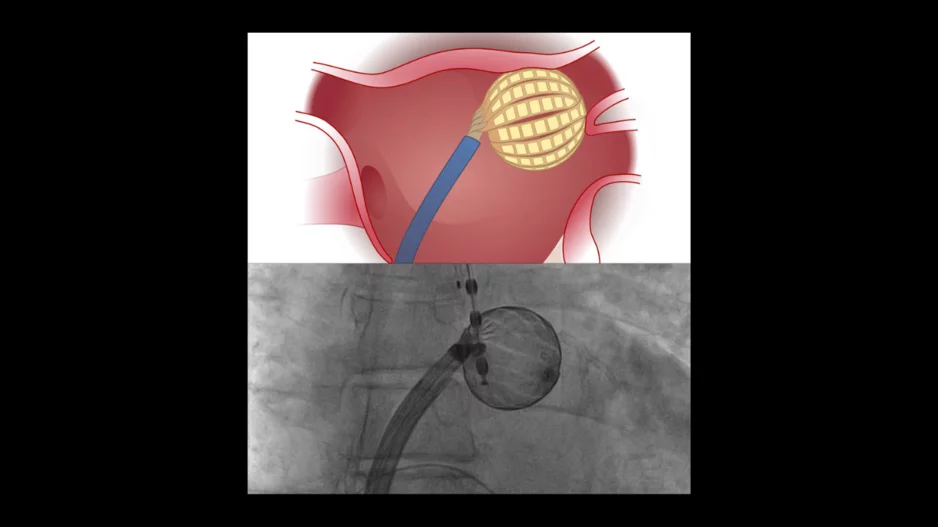
A new pulsed field ablation (PFA) system from Kardium could give cardiologists a new tool in the war against atrial fibrillation (AFib), according to new data published in Heart Rhythm.[1]
The…
WearLinq, a San Francisco-based healthcare technology company, has acquired AMI Cardiac Monitoring, an independent diagnostic testing facility, to help increase the reach of its…
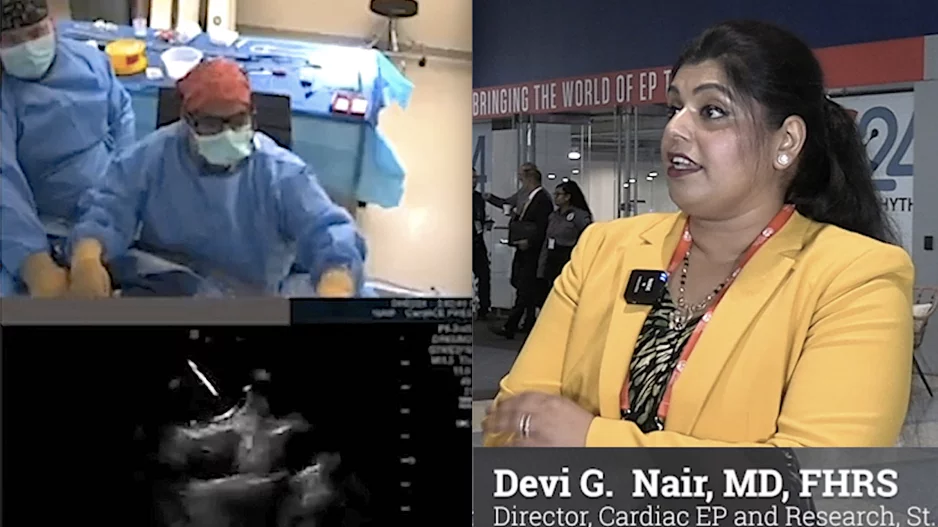
The late-breaking U.S. multicenter admIRE clinical trial at the Heart Rhythm Society 2024 meeting, showed positive data on the long-term safety and effectiveness of the the Biosense Webster Varipulse…