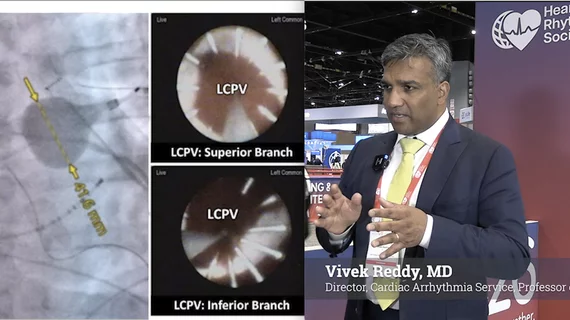
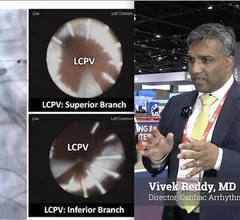
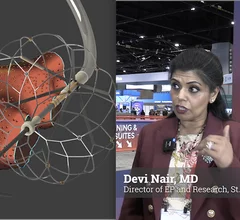
Heart Rhythm Society (HRS)
The Heart Rhythm Society (HRS) is the primary U.S. medical society for electrophysiology (EP). The organization promotes education and advocacy for cardiac arrhythmia professionals and patients, including setting policy guidelines and offering expert consensus.
Displaying 1 - 8 of 104