More Articles
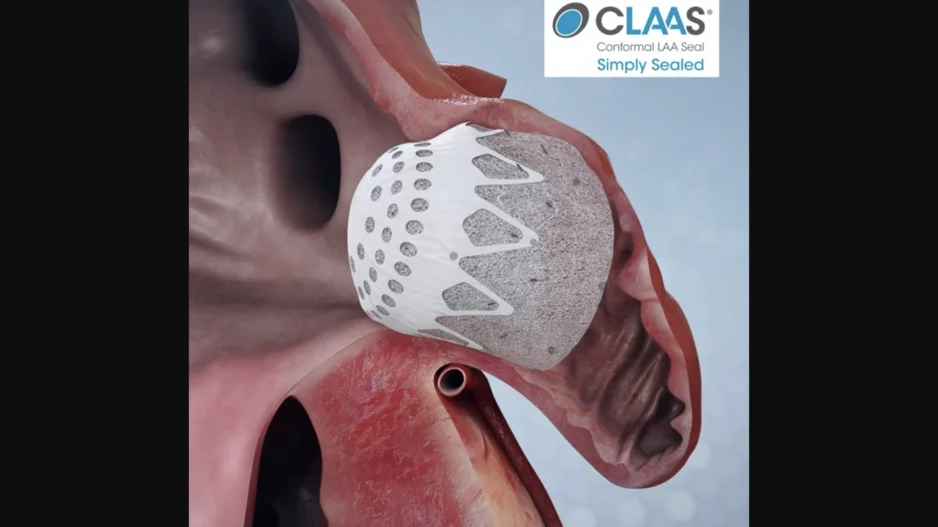
W. L. Gore & Associates, a global materials science company with headquarters in Delaware, has finalized its acquisition of Conformal Medical, the New Hampshire-based medical device company behind the…
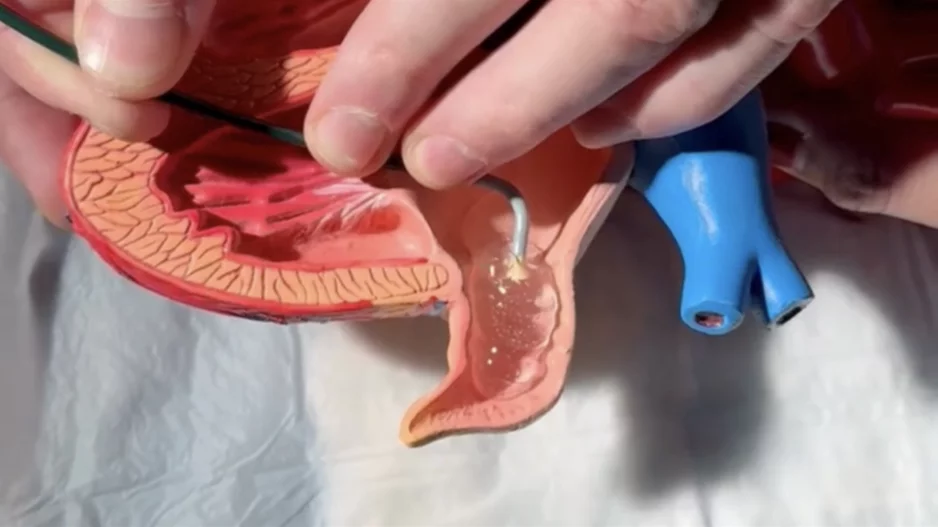
After years of implanting left atrial appendage occlusion (LAAO) devices in atrial fibrillation (AFib) patients, an interventional cardiologist with the University of Chicago Medical Center (UChicago…
Medtronic reported $9.02 billion in sales for its third quarter, up 8.7% compared to the previous year. That figure includes $3.45 billion in sales for the company’s cardiovascular portfolio, good for an increase of 13.8%.
The success…
The U.S. Food and Drug Administration (FDA) granted 510(k) clearance for the HeartBeam 12-lead electrocardiogram (ECG) synthesis software for the home assessment of…
Legislative mandates requiring all competitive athletes to undergo electrocardiogram (ECG) screening will likely do more harm than good, according to a new statement from the American College of Cardiology (ACC)…
BioCardia, a California-based regenerative medicine company, is closer than ever to receiving U.S. Food and Drug Administration (FDA) approval for a new catheter capable of delivering cell and gene therapies…
Medline Industries, an Illinois-based manufacturer and distributor of medical supplies, is recalling specific lots of several electrophysiology (EP) catheters due to significant health risks. The devices, originally made by a variety vendors, may…
The world of cardiovascular diagnostics has evolved quickly in recent years, making the leap from traditional electrocardiogram (ECG) systems and…
The Heart Rhythm Society (HRS) has published additional details to help its member manage an ongoing safety concern with hundreds of thousands of Boston Scientific defibrillation leads.
While a…
The 31st Annual International AF Symposium in Boston included late-breaking presentations from many of the leading names in …
Milestone Pharmaceuticals said it plans to begin U.S. commercialization of its etripamil (Cardamyst) nasal spray, indicated for the conversion of acute symptomatic episodes of paroxysmal supraventricular…
eMurmur, an Ontario-based artificial intelligence (AI) startup, has received U.S. Food and Drug Administration (FDA) clearance for its…
Medtronic has secured CE mark approval for its Affera Sphere-360 catheter, an all-in-one mapping and single-shot pulsed…
Abbott reported $11.5 billion in sales for the fourth quarter of 2025, a 4.4% increase compared to 2024 that translates to $1.01 per share. While this just barely missed the predictions of some analysts, the company did see its medtech sales grow…
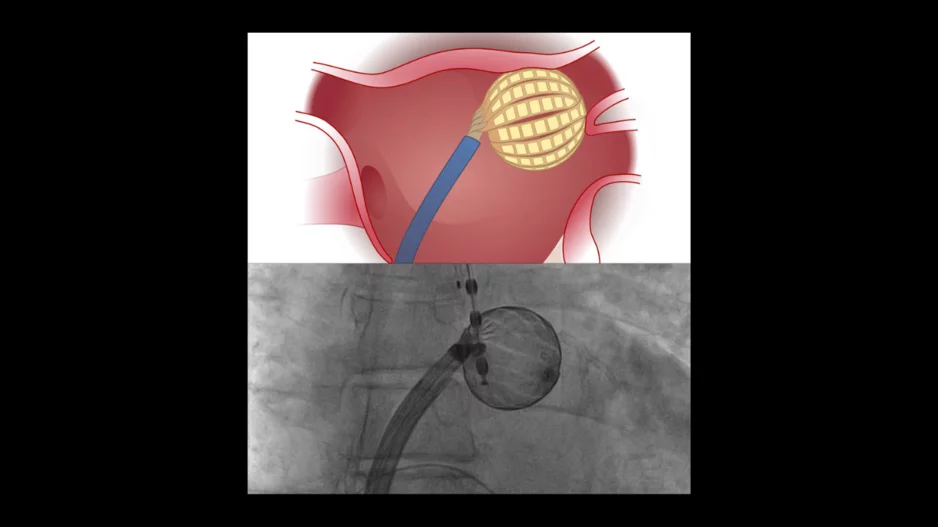
The Globe Pulsed Field System, a new pulsed field ablation (PFA) system from Vancouver-based Kardium, is a safe, effective treatment option for paroxysmal atrial fibrillation (AFib), according to new findings…
Abbott is partnering with California-based AtaCor Medical to develop a next-generation extravascular implantable…
Abbott has received CE mark approval for its TactiFlex Duo Ablation Catheter, Sensor Enabled for the treatment of atrial fibrillation (AFib).
The device was designed to deliver both pulsed field ablation (PFA) energy and…
Imricor Medical Systems, a Minneapolis-based medtech company, has received U.S. Food and Drug Administration (FDA) clearance for its Vision-MR…
Boston Scientific has received U.S. Food and Drug Administration (FDA) approval for its…
AccurKardia, a New York-based medtech company, has received U.S. Food and Drug Administration (FDA) clearance for the latest version of its AccurECG Analysis Software…