More Articles
Social isolation and loneliness are both independently linked to a heightened risk of developing heart failure, according to new research published in …
The U.S. Food and Drug Administration (FDA) kicked off 2023 with a busy January. The agency approved a new…
The U.S. Food and Drug Administration (FDA) has announced that Datascope, a subsidiary of Getinge, is recalling two different Cardiosave intra-aortic balloon pumps (IABPs) due to a risk of blood entering…
The most common causes of mortality during transvenous lead removal (TLR) in patients with cardiac implantable electronic devices (CIEDs) are infection and decompensated heart failure, according to new research published in JACC:…
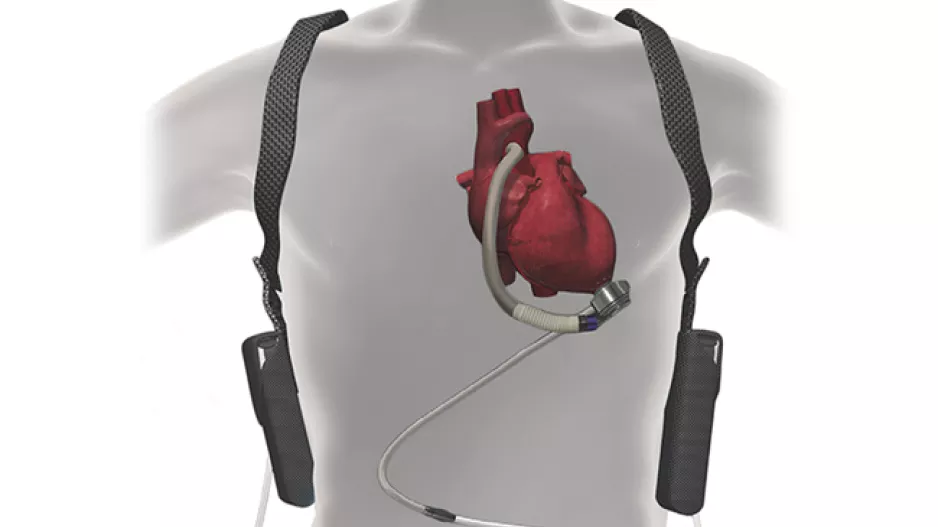
When Medicare updated its policy on left ventricular assist devices (LVADs) in 2020, making it easier for heart failure patients to receive an LVAD at a…
Rehospitalization after aortic valve replacement (AVR) is associated with an increased risk of patient mortality, according to new findings published in the Circulation: Cardiovascular Interventions.[1] This suggests that…
Regulatory Roundup: FDA news on omecamtiv mecarbil for HFrEF, 'world's smallest heart pump' and more
December was another big month for the U.S. Food and Drug Administration (FDA). The agency…
Anumana, a Massachusetts-based healthcare technology company focused on artificial intelligence (AI), has announced a new multi-year deal with Pfizer…
Some cancer therapies—primarily those that attack a patient’s DNA—have been linked to cardiovascular risks such as cardiac toxicity and heart failure. The…
Specialists with the Albert Einstein College of Medicine at Montefiore Health System have received a five-year grant worth $5.2 million from the…
Ultromics, a U.K.-based healthcare technology company focused on heart failure detection, has received U.S. Food and Drug Administration (FDA) clearance…
Heart failure patients often treat their symptoms with alternative therapies and over-the-counter supplements. While some of these treatments may prove…
The U.S. Food and Drug Administration (FDA) has shared updated recommendations for the use of Abiomed’s Impella RP System after reviewing final post-approval study (PAS) results. Those…
The U.S. Food and Drug Administration (FDA) has shared the details of another recall associated with Medtronic’s HeartWare Ventricular Assist Device (HVAD) system.
…
Heart failure patients are less likely to fill prescriptions for beneficial medications if they have a high copay, according to new data published in the Journal of the American Heart Association.[1]
…
Regularly adding a little more salt to your meals may be having a significant impact on your risk of developing cardiovascular disease (CVD) or heart failure…
Artificial intelligence (AI) can be applied to …
Two heart specialists at Hackensack University Medical Center (HUMC) in Hackensack, New Jersey, became the first surgeons in the…
Flu shots are associated with a significant reduction in the risk of cardiovascular complications or pneumonia in patients with heart failure, according to…
The American Heart Association (AHA) has announced 35 late-breaking studies and 42 featured science sessions being presented at the 2022 annual meeting in Chicago, Nov. 5-7.












![Salt substitutes are associated with a reduced risk of high blood pressure without increasing the risk of low blood pressure, according to new findings published in the Journal of the American College of Cardiology.[1]](/sites/default/files/styles/media_image/public/2018-04/salt-3285024_1920.jpg.webp?itok=m51O2CIk)