More Articles
Treatment with dapagliflozin is associated with improved symptoms in patients with heart failure with reduced ejection fraction (HFrEF), but not patients with heart failure with preserved ejection fraction (HFpEF), according to new…
BiVACOR, an international medical device company with headquarters in the United States and Australia, has received approval from the U.S. Food and Drug Administration (FDA) to kick off an…
Larry Allen, MD, a cardiologist with the University of Colorado School of Medicine, has received $7 million in…
Regular marijuana use may significant increase a person’s risk of developing heart failure, according to new research presented at the…
Weekly treatments of semaglutide can reduce the risk of cardiovascular death, heart attack or stroke among obese patients without diabetes, according to new data presented at the…
The use of artificial intelligence (AI) to evaluate and treat heart disease has been one of the biggest stories at the…
When the Centers for Medicare and Medicaid Services (CMS) finalized the…
CVRx, a Minneapolis-based medical device company focused on heart failure treatments, is celebrating a key update the …
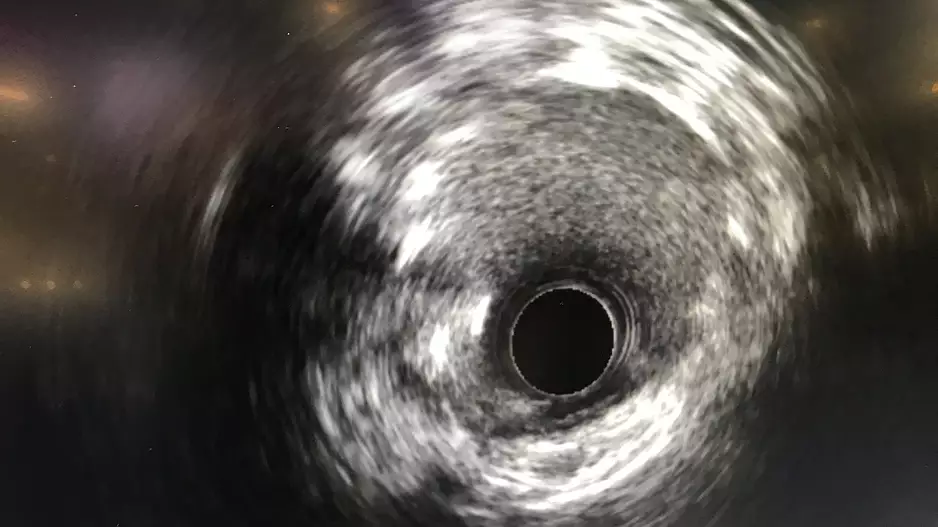
Using intravascular ultrasound (IVUS) to guide percutaneous coronary intervention (PCI) procedures is associated with a significant improvement in survival after two years, according to new findings published in JSCAI, the…
Lawrence Faucette, the second patient to ever receive a genetically modified pig heart, died on Oct. 30. He was 58 years old.
Faucette…
Ventricle Health, a North Carolina-based virtual cardiology clinic backed by private-equity investors, has raised $8 million to fund the expansion of its heart…
Dihydropyridines, a group of calcium channel blockers commonly prescribed to treat hypertension, have been the topic of some debate in recent months due to…
The U.S. Food and Drug Administration (FDA) has issued a new warning letter to Abiomed, a Johnson and Johnson company, describing a variety of “significant violations” identified by the agency’s…
Endotronix, an Illinois-based healthcare technology company focused on developing heart failure treatments, gained an investigational device exemption (IDE…
Treatment with cannabidiol (CBD) may be beneficial for patients with heart failure with preserved ejection fraction (HFpEF), according to a new study…
Alnylam Pharmaceuticals has announced that the U.S. Food and Drug Administration (FDA) rejected its request for patisiran to be approved to treat cardiomyopathy from hereditary transthyretin-mediated…
The American Heart Association (AHA) has developed a brand new strategy for the prevention and management of cardiovascular disease (CVD).
This updated approach…
Artificial intelligence (AI) is being developed to help make cardiovascular clinical trials more efficient, lead to faster advances in patient care, and bring…
Anumana, a Massachusetts-based nference portfolio company, has received U.S. Food and Drug Administration (FDA) clearance for its new…
Patients with cannabis use disorder (CUD) face a heightened risk of adverse cardiovascular events, according to new data published in Addiction.[1]
“Mounting epidemiological, clinical and…
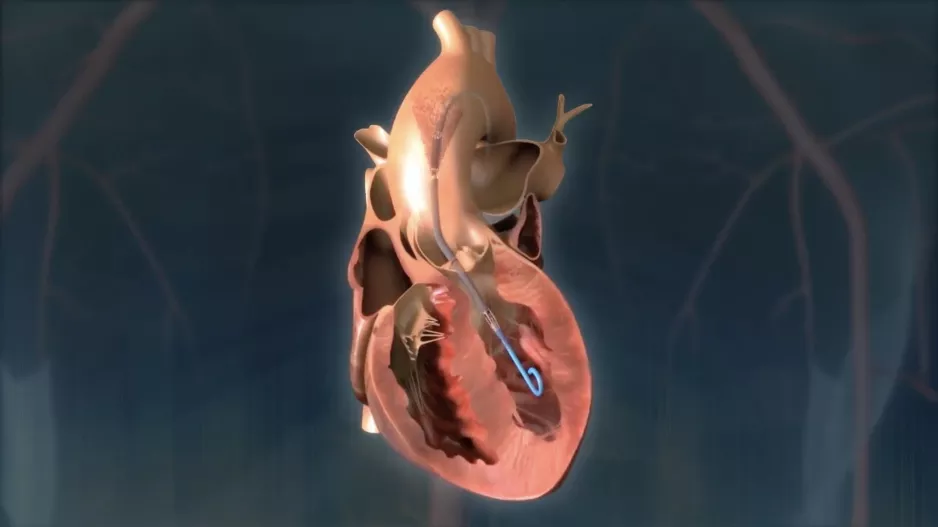
Nearly one-fifth of patients die within two years of undergoing transcatheter aortic valve replacement (TAVR), according to a new analysis…