Clinical
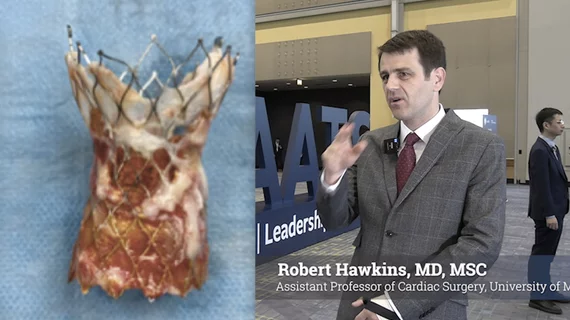
This channel newsfeed includes clinical content on treating patients or the clinical implications in a variety of cardiac subspecialties and disease states. The channel includes news on cardiac surgery, interventional cardiology, heart failure, electrophysiology, hypertension, structural heart disease, use of pharmaceuticals, and COVID-19.
Displaying 1 - 8 of 7388